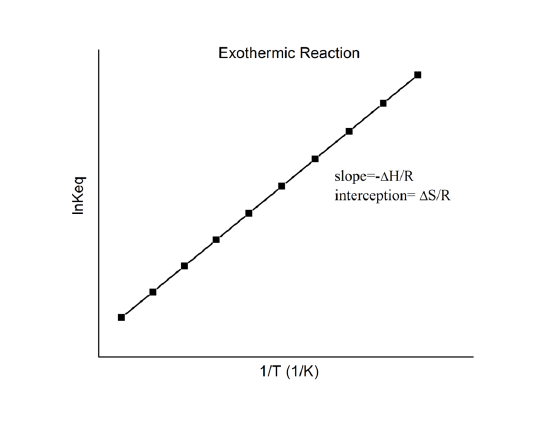
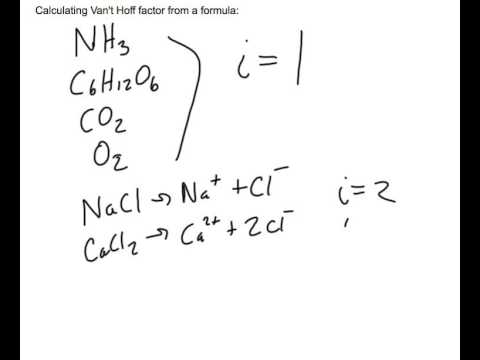
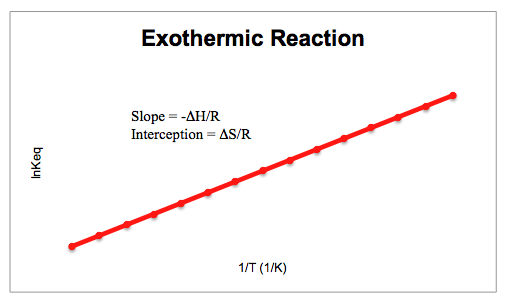
The Van't Hoff factor of 0.1 M {text{Ba}}{left( {{text{N}}{{text{O}}_3}} right)_2} solutions is 2.74. The degree of dissociation will be 91.3%74%87%100%

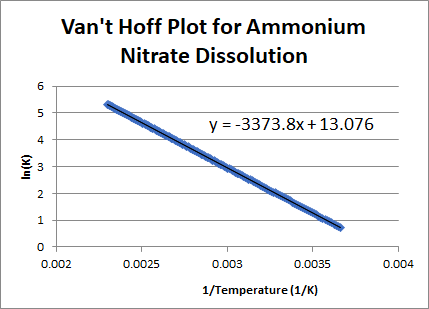
Plot of Van't Hoff equation for adsorption of Cr(VI) on to MPGP. The... | Download Scientific Diagram