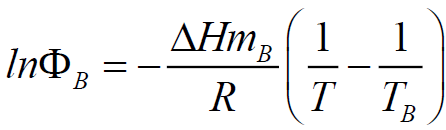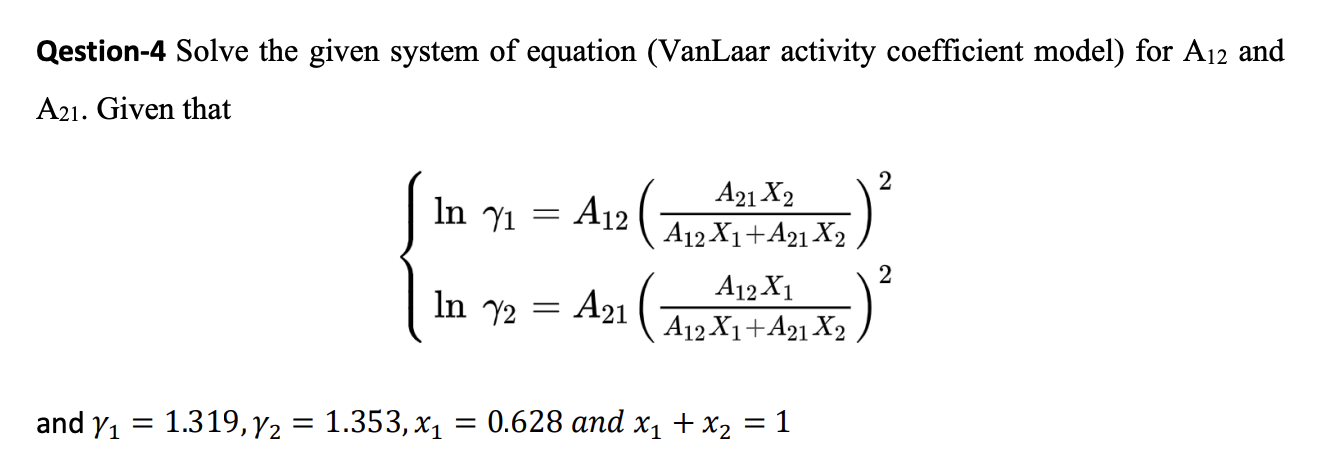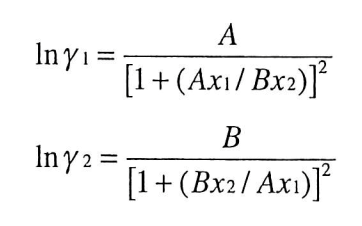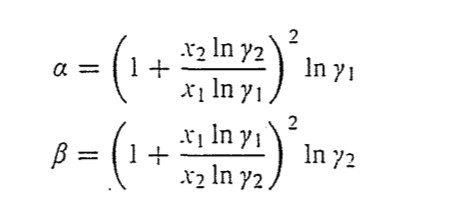SciELO - Brasil - Measurement and correlation of isobaric vapor-liquid equilibrium for the binary system of cyclopentane and tetrahydrofuran Measurement and correlation of isobaric vapor-liquid equilibrium for the binary system of cyclopentane

Optimum binary interaction parameter and van Laar con- stants in the... | Download Scientific Diagram
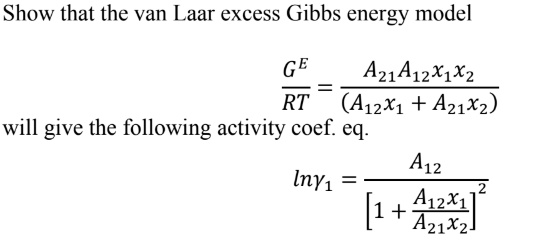
SOLVED: Show that the van Laar excess Gibbs energy model GE = A12x1x2RT / (A12x1 + A21x2) will give the following activity coefficient equation: A12lny1 = 1 / (A12x1 + A21x2)

Figure 7 from Error analysis of linearization methods in regression of data for the Van Laar and Margules equations | Semantic Scholar
DETERMINATION OF COMPOSITION-DEPENDENI' LIQUID ACTIVITY COEFFICIENrS1 BY USE OF THE VAN LAAR EQUATION GARRY LEONARD PIGG MASTER