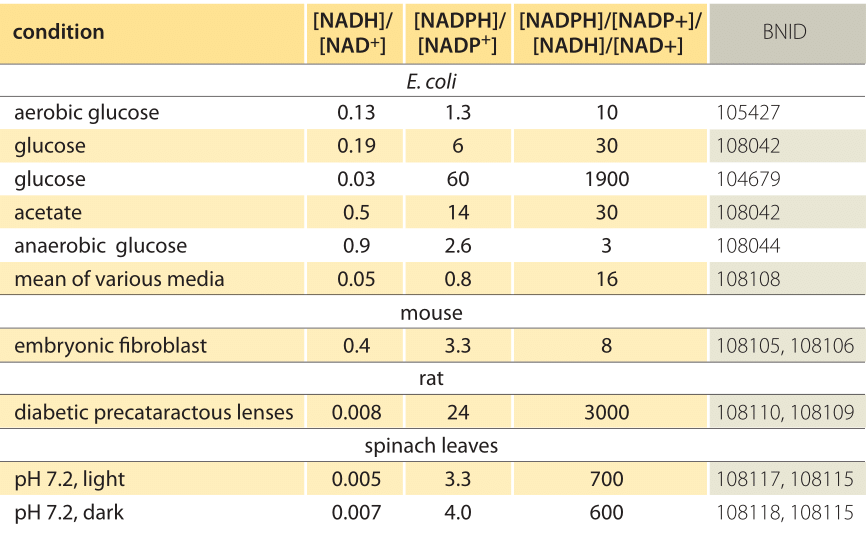
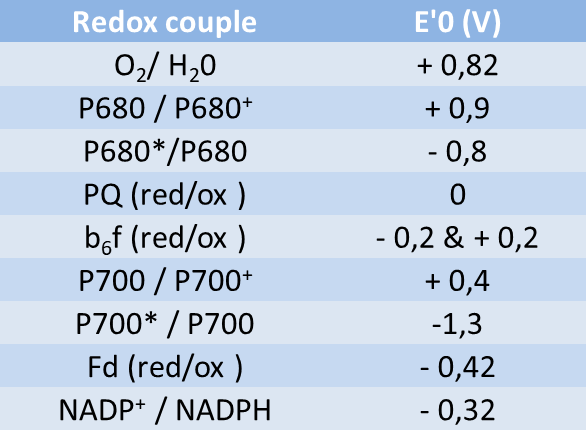
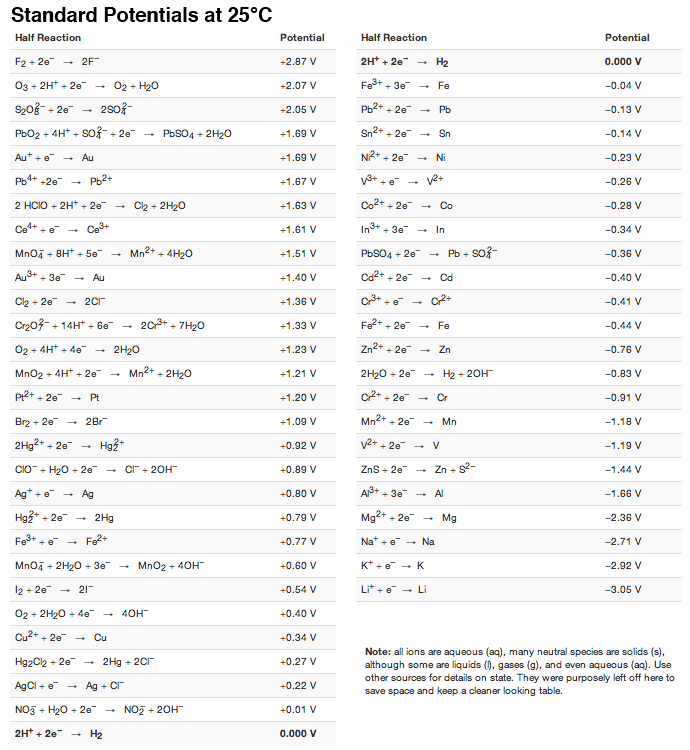
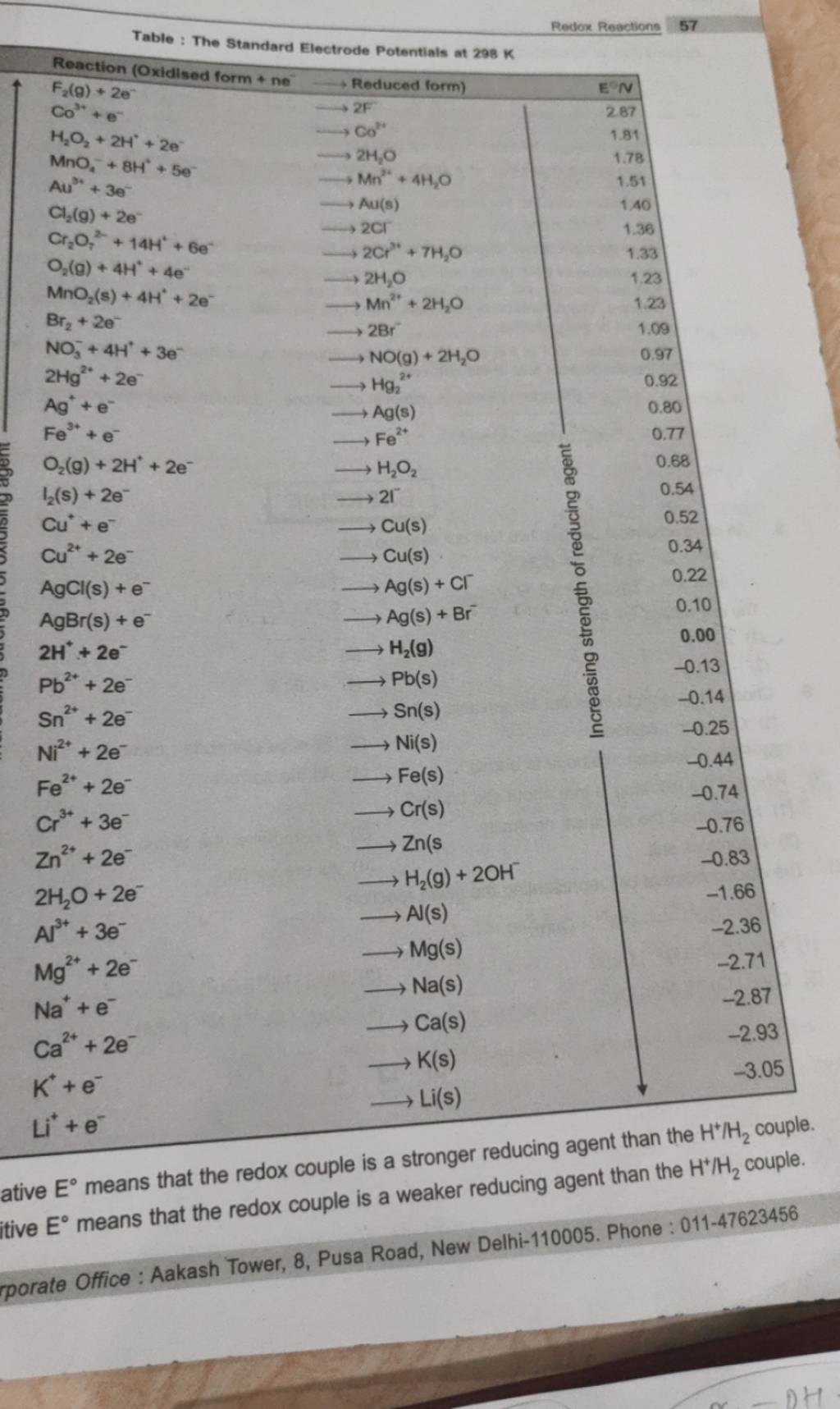
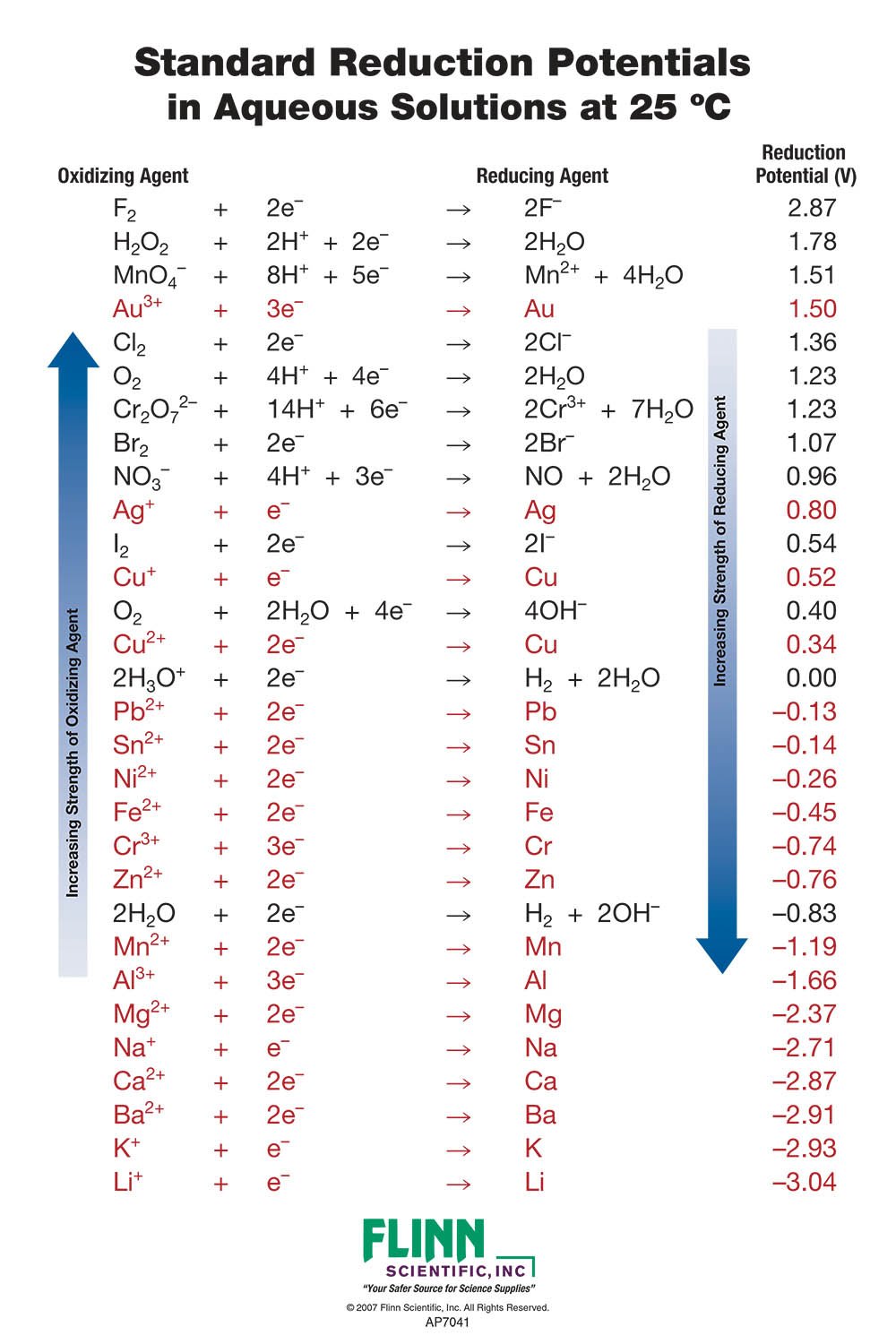
Using the standard electrode potentials given in Table 3.1, predict the reaction between the following is feasible:(i) Fe^{3+}(aq) and I^{-}(aq)(ii) Ag^{+} (aq) and Cu(s)(iii) Fe^{3+} (aq) and Br^{-} (aq)(iv) Ag(s) and Fe^{3+} (
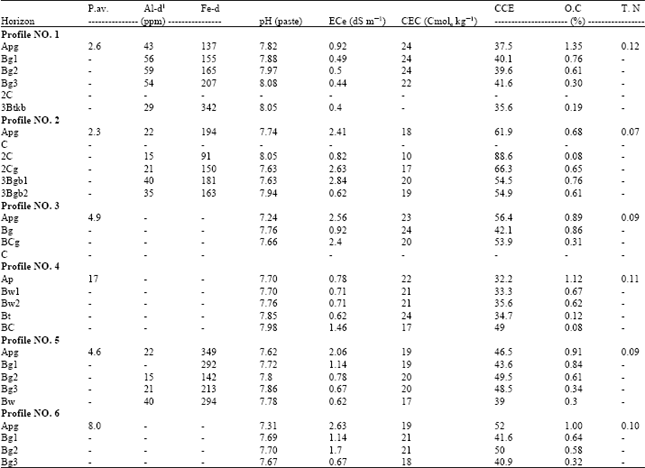
Study of Reduction-Oxidation Potential and Chemical Charcteristics of a Paddy Field During Rice Growing Season














![Redox Potential Values (in volts vs Fc + /Fc) of Borane [B 12 H 12 ] 2−... | Download Table Redox Potential Values (in volts vs Fc + /Fc) of Borane [B 12 H 12 ] 2−... | Download Table](https://www.researchgate.net/publication/310382562/figure/tbl1/AS:668992016498699@1536511534809/Redox-Potential-Values-in-volts-vs-Fc-Fc-of-Borane-B-12-H-12-2-and-Its.png)
![AUFBAU1 [REFERENCE SECTION: REDOX POTENTIALS] AUFBAU1 [REFERENCE SECTION: REDOX POTENTIALS]](https://www.wissensdrang.com/media/tablerp.gif)