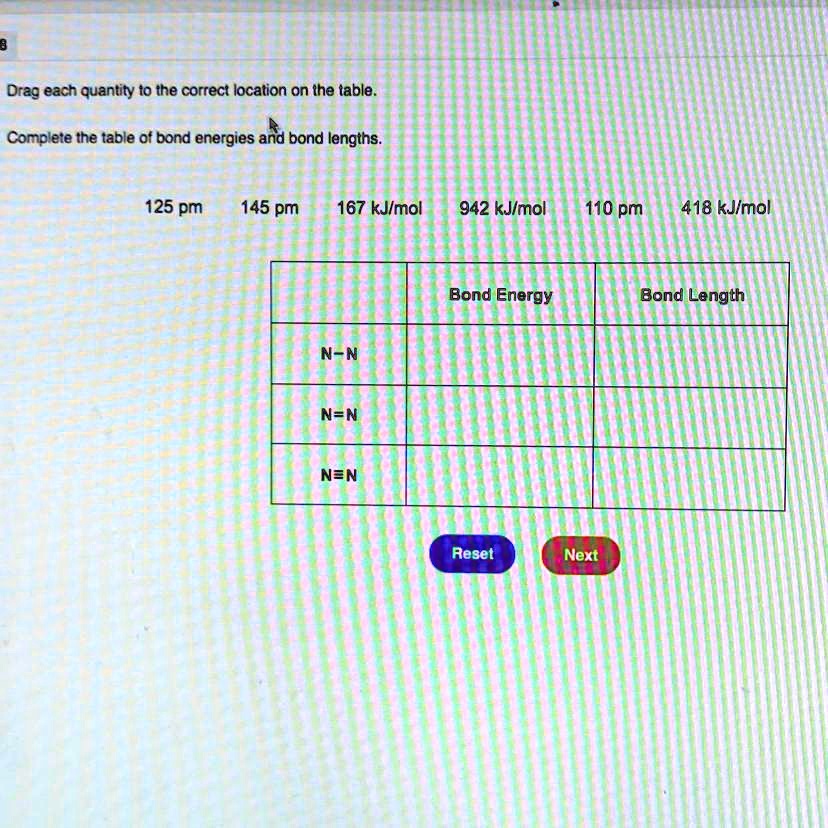
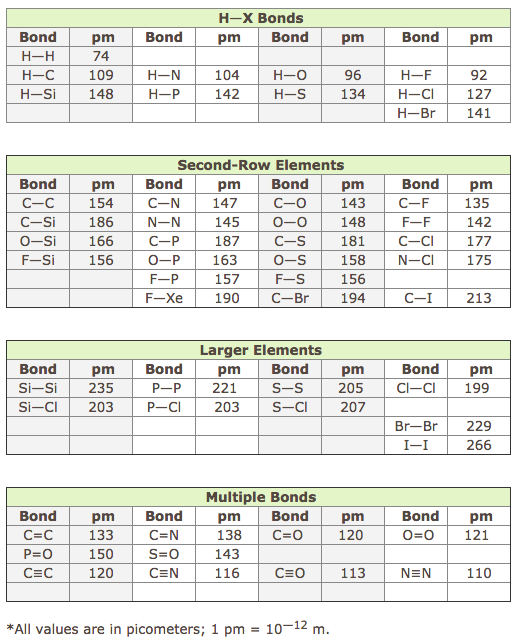
![PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/229ac8099c4a475d47a8b557fd14326f02189409/4-Table1-1.png)
PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar
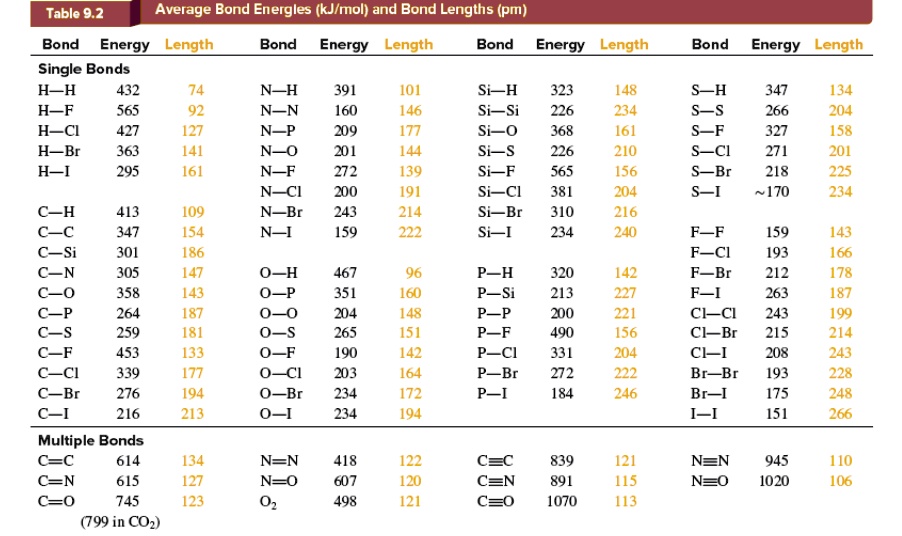
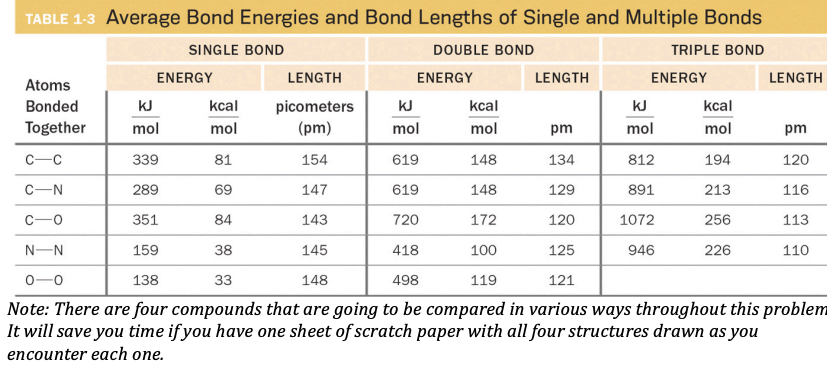
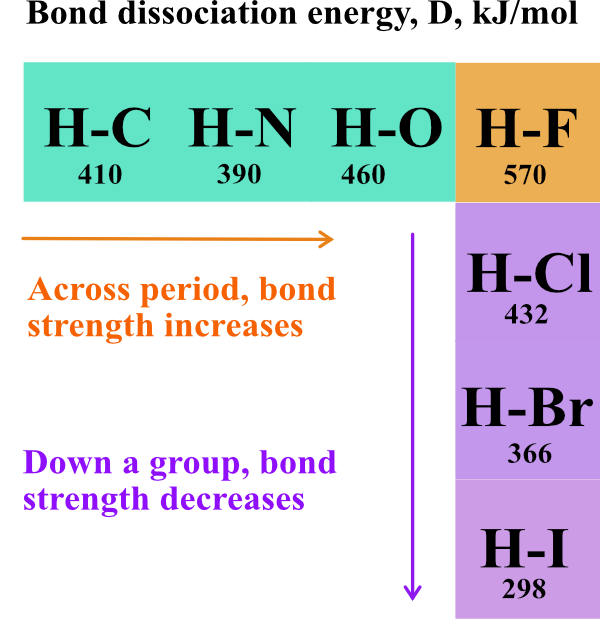
SOLVED: Table 9.2 Average Bond Energies (kJ/mol) And Bond Lengths (pm) Bond Energy Length Single Bonds H-H 432 H-F 565 H-Cl 427 H-Br 363 H-I 295 Bond Energy Length N-H 391 N-N

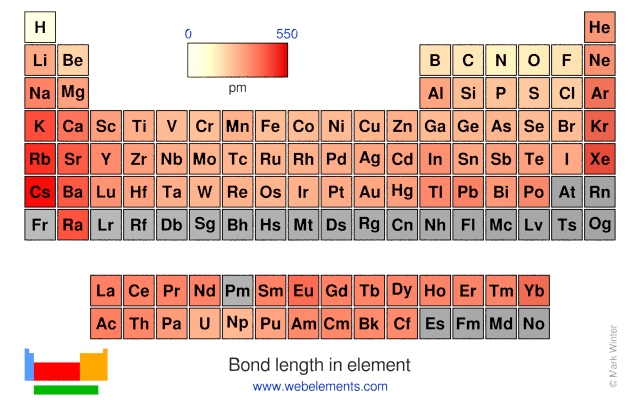
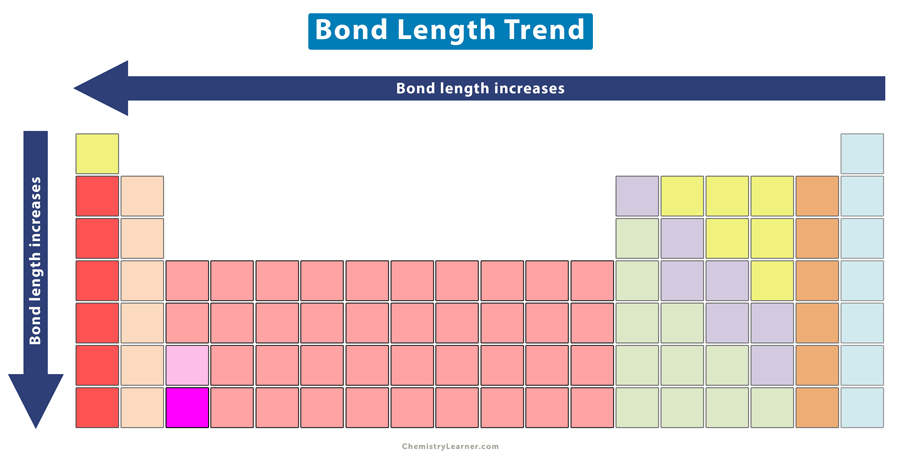
In the given table as we move downward, the group members become larger. Due to this, is there any indication that there is a trend of bond lengths becoming longer? | Homework.Study.com
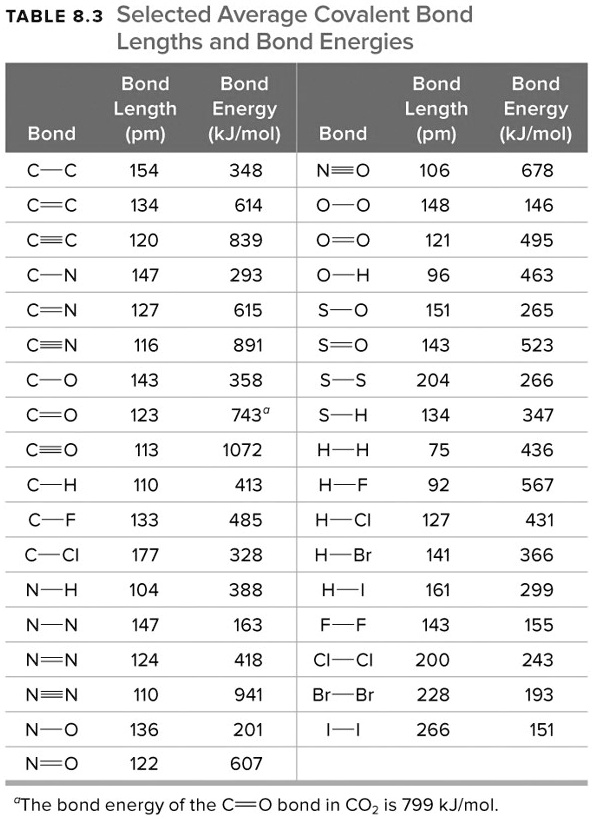
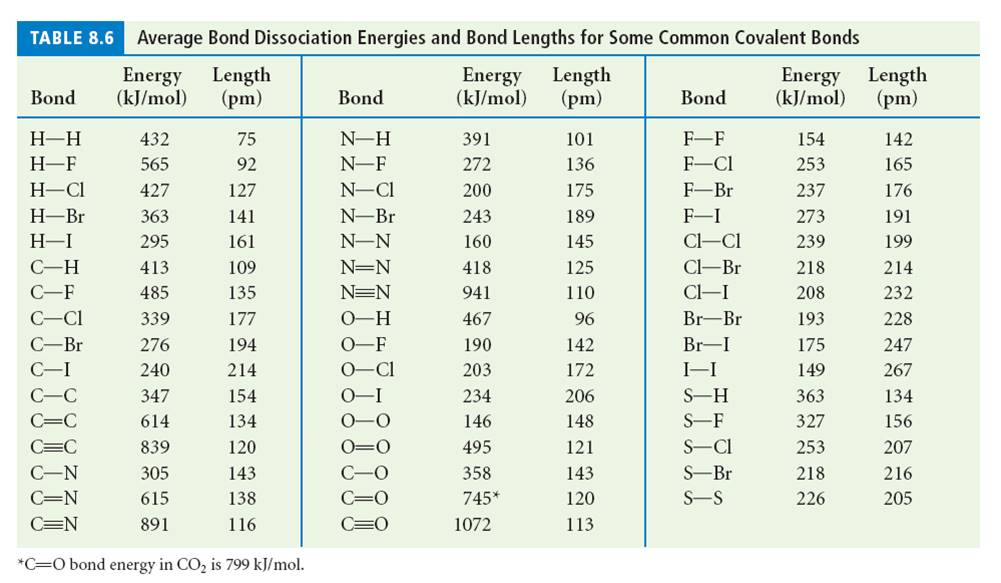
SOLVED: TABLE 8.3 Selected Average Covalent Bond Lengths and Bond Energies Bond Length (pm) Bond Energy (kJ/mol) C-C 348 C=C 106 678 C=N 154 N=O 134 C=O 614 C-F 148 C-Cl 146

Table 1. X-ray and DFT calculated bond lengths () of (5) : Synthesis, XRay Crystallography and DFT Studies of Ni(II) Complex with Tetradentate : Science and Education Publishing