N2O conversion over Ir(K)/Al2O3 catalysts in the presence of O2 (GHSV:... | Download Scientific Diagram

N2 conversion in the coaxial DBD reactor packed with beads of γ-Al2O3,... | Download Scientific Diagram

Exploration and tuning of Al2O3/Mo composite for enhancement of anti-corrosion and tribological characteristics in zinc phosphate conversion coatings - ScienceDirect
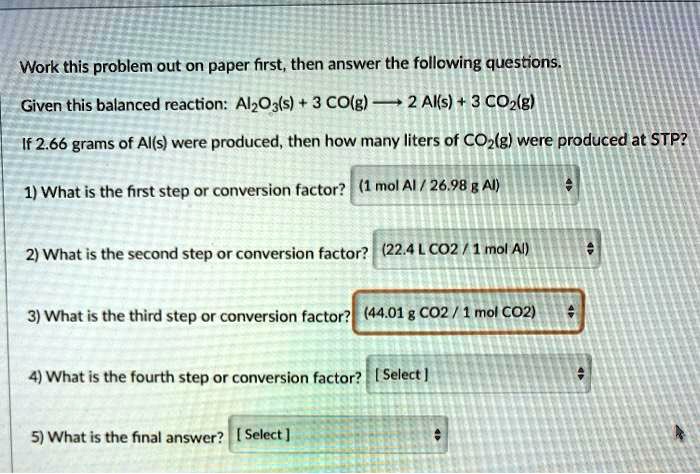
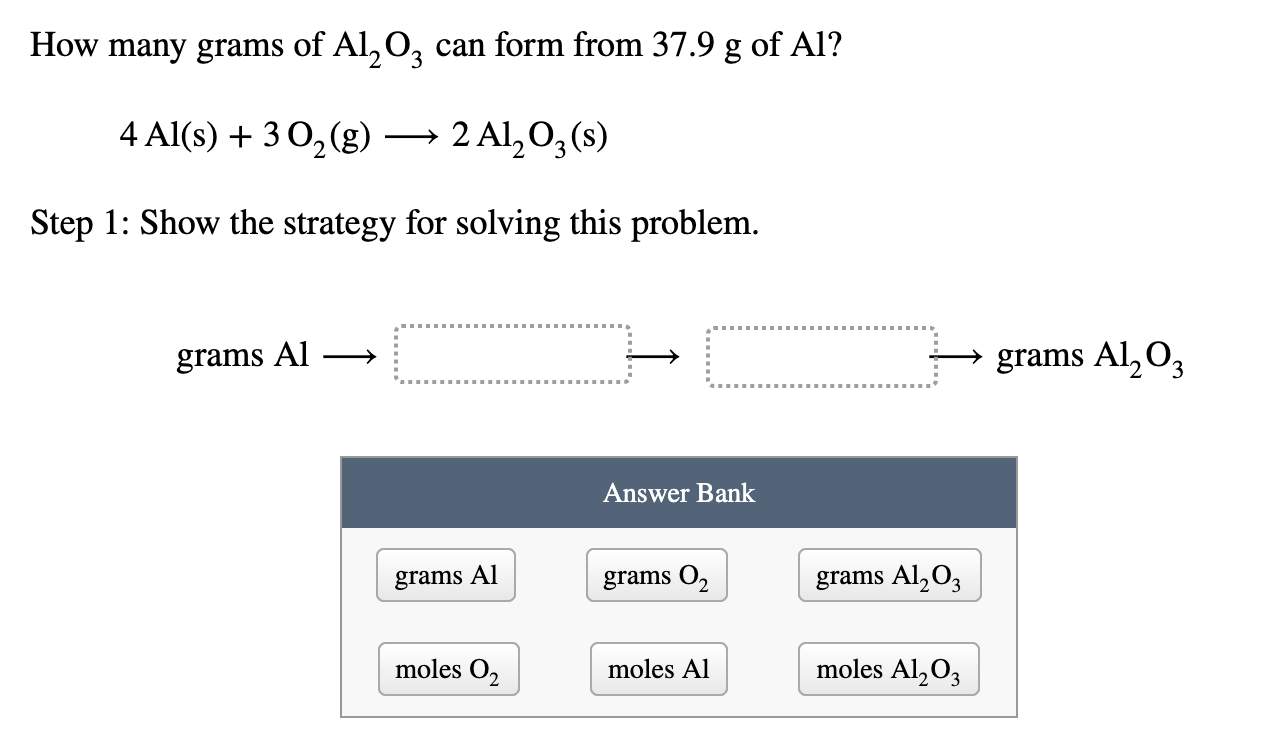
SOLVED: Identify the conversion steps needed to calculate the formula units of aluminum oxide, Al2O3, formed by the following reaction if 7.2×1021 molecules of oxygen, O2, react. 4 Al (s)+3 O2 (g)→2

a) 1,2-DCE conversion curves of NM/γ-Al2O3 catalysts; (b) Arrhenius... | Download Scientific Diagram

Reaction rate (%) of Al 2 O 3 and formation rate (%) of h-AlN and c-AlN... | Download Scientific Diagram

NH 3 conversion in the NH 3 -SCO reaction over Pt/Al 2 O 3 catalysts in... | Download Scientific Diagram

CO conversion vs temperature plots for Co/γ-Al2O3 samples, prepared... | Download Scientific Diagram

CO conversions as a function of reaction temperature over Au/ Al 2 O 3... | Download Scientific Diagram

SOLVED: The following equation shows the conversion of aluminum oxide (from the ore bauxite) to aluminum: 2 Al2O3 (s) â†' 4 Al (s) + 3 O2 (g) ΔH = -801 kcal Is